Quality and Safety

Kirin Group’s Quality Assurance Policies and Approaches

Yoshiyuki Yonetani
Senior Executive Officer
Chief Quality Assurance Officer
The goal of the Kirin Group’s quality assurance activities is to provide consumers and patients with products and services that are safe and reliable.
To achieve this, we place the highest priority on ensuring safety and delivering consumer and patient satisfaction, in accordance with the Kirin Group’s core values of “Pioneer with Innovation”, “Consumer/Patient at Heart” and “Quality in Mind”.
These principles are embodied in the Kirin Group’s Quality Policy and the Commitments to Action, which guide our efforts across all business domains—Alcoholic Beverages, Non-alcoholic Beverages & Health Science, and Pharmaceuticals—in Japan and overseas, and are reflected in our daily quality assurance practices.
Kirin Group’s Quality Policy
Guided by our fundamental Values in the KIRIN WAY — “Pioneer with Innovation”, "Consumer/Patient at Heart" and “Quality in Mind"— Kirin Group places the satisfaction of our consumers and patients, and the delivery of safe products and services that give them peace of mind, above all else.
Commitments to Action
1.Improving Consumer/Patient satisfaction
We act with genuine care and integrity toward our consumers and patients. We listen closely to their voices, using their feedback to continuously improve quality, create meaningful value, and make thoughtful, detailed improvements that enhance consumer and patient satisfaction.
2.Safety, Reliability and Trust
From sourcing raw materials and supplies to development, manufacturing, testing, logistics, and sales, we prioritize quality at every step to deliver safe products and services that give consumers and patients confidence and peace of mind.
3.Compliance
We comply with all applicable laws and regulations and hold ourselves to high voluntary standards.
4.Provision of accurate quality information
We are committed to providing accurate, transparent information that fosters trust with our consumers and patients.
5.Objective and recognized quality-assurance practices
We will implement and maintain a world-class, accredited quality assurance system to ensure the highest standards.
6.Continuous improvement
We will develop and acquire advanced technologies, invest in our people, and continuously enhance and evolve our quality assurance efforts.
Revised date : Feb. 13th, 2026
Takeshi Minakata
Representative Director of the Board,
President & COO
Kirin Holdings Company, Limited
Our Quality Management
Kirin Group has established the Kirin Group Global Quality Management Principle, which embodies the Kirin Group’s Quality Policy and the Commitments to Action to promote a high level of quality management by Kirin Group companies.
Quality Assurance Approaches
We take the following approaches in our businesses to ensure the safety and reliability of our products and services, and to retain the trust and satisfaction of consumers and patients.
Our Approaches in the Alcoholic Beverages & Non-alcoholic Beverages Business
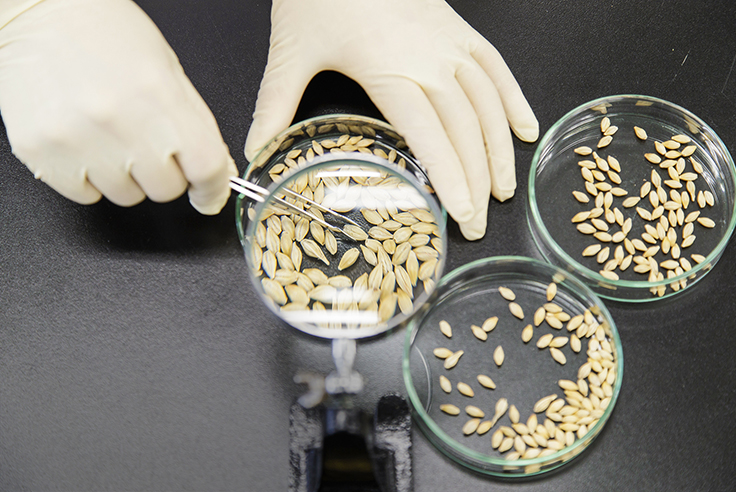
We are committed to meeting the stringent quality control targets established across the value chain in order to continue to deliver safe, reliable, and high-quality products and services to consumer. We use internationally recognized standards for quality assurance to audit and evaluate our quality assurance practices and performance and continually improve them.
Our Approaches in the Health Science Business
Group companies manufacture and market Foods with Function Claims and Food for Specified Health Uses, using proprietary materials developed by our research laboratories.
To deliver the safe and reliable products and services that consumer expect from Kirin Group, we have combined the best of the quality assurance system established through years of manufacturing alcoholic beverages and non-alcoholic beverages and the dependability assurance system modeled after the one used in the Pharmaceuticals business.

Our Approaches in the Pharmaceuticals Business

Kirin’s Quality Assurance in the Pharmaceuticals Business
We follow the Good Manufacturing Practice (GMP) and other GxP* guidelines at every step from research and development to manufacturing and marketing to after-sale follow-ups of pharmaceutical products in order to ensure their quality as well as their safety for users, enables us to continue to deliver products with proven effectiveness and safety consistently.
-
*GxP (Good [variable] Practice) is a set of regulations and guidelines formulated to ensure the effectiveness, safety, and quality of pharmaceutical products throughout every stage from product development to after-sales follow-ups.