Ensuring Product Safety and Reliability in the Health Science Business
Our Quality Assurance Practices in the Health Science Business in Japan
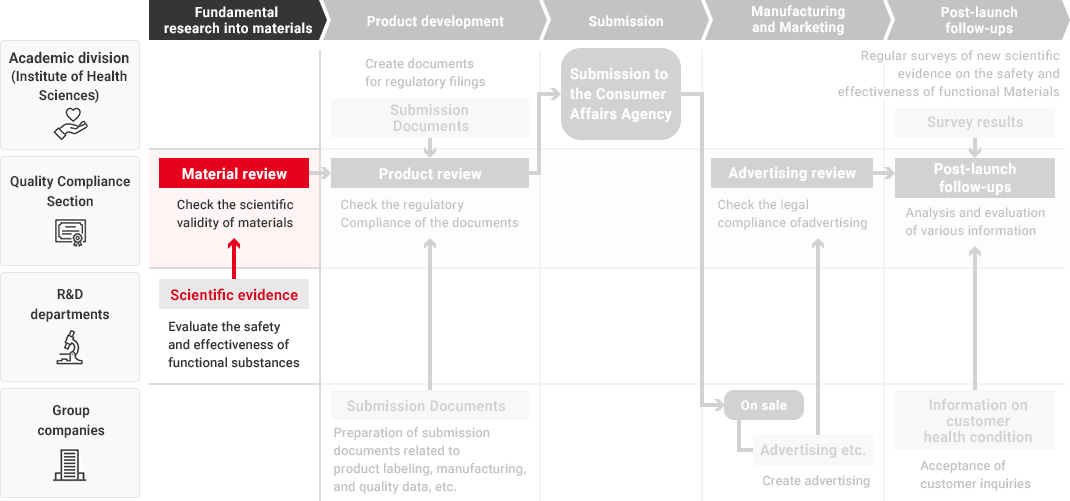
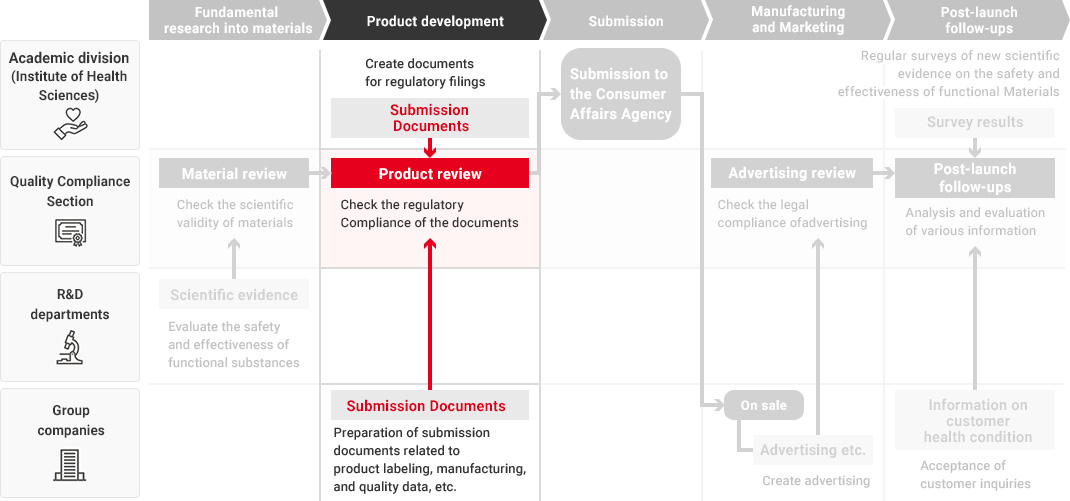
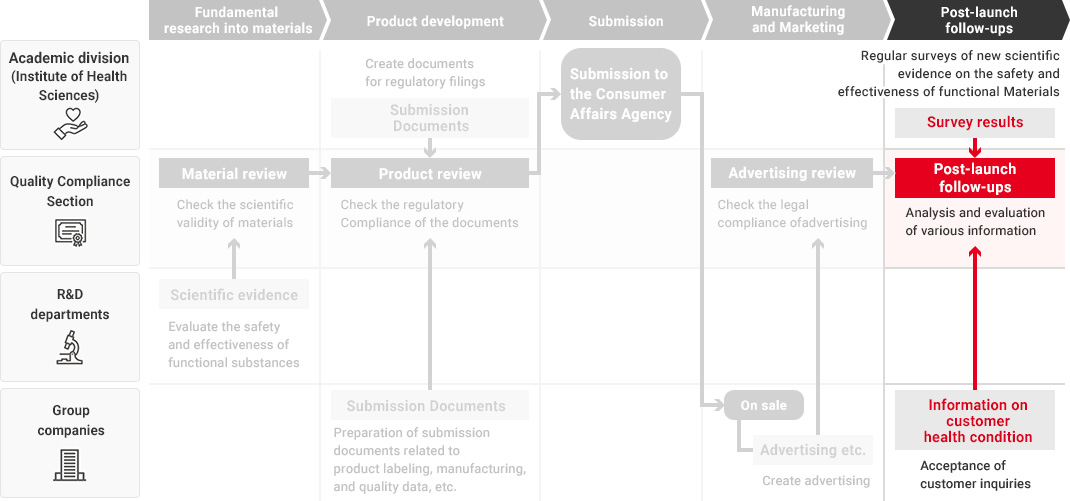
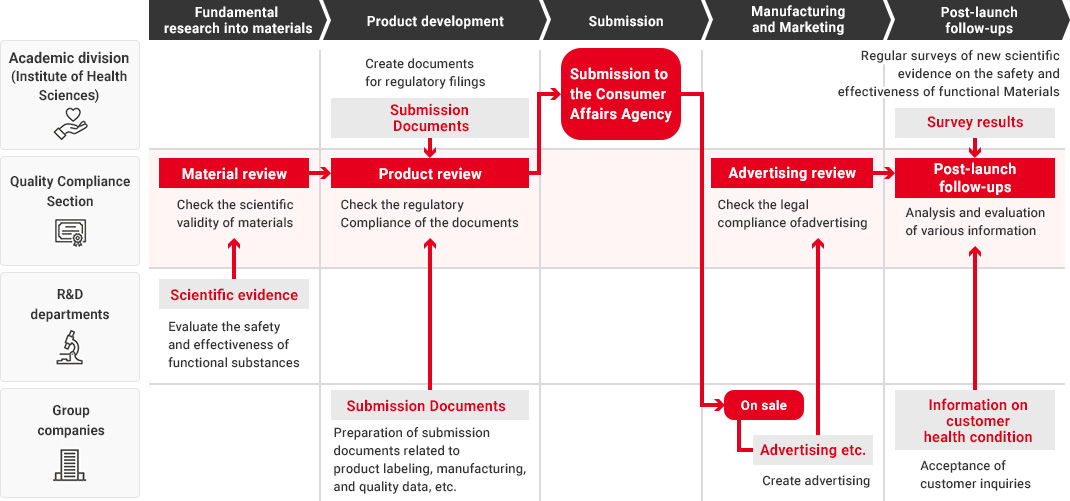
In the Health Science business, our Research and Development Department, Quality Compliance Section, the academic division, and Kirin Group companies that manufacture and market health science products all work together in promoting quality assurance that starts from fundamental research into materials and ingredients and extends to product development and post-launch follow-ups.
We leverage technologies accumulated in our alcoholic beverages, non-alcoholic beverages and pharmaceuticals businesses as well as our wealth of advanced fermentation and bio technologies to conduct a wide range of fundamental research into materials and ingredients. When developing new health science products, each Group company utilizes the developed materials and conducts quality assessments in a manner tailored to each product type. During this process, the Quality Compliance Section evaluates the safety and effectiveness of materials and products across the value chain as part of its efforts to implement upgraded quality assurance practices in the Health Science business.

Initiatives in Foods with Function Claims
Here we introduce our initiatives for safety and reliability in the Health Science business, using Foods with Function Claims as an example.
Foods with Function Claims
Under the food business operator's own responsibility, Foods with Function Claims can be labeled with function claims based on scientific evidence. Information on the evidence supporting the safety and effectiveness of the product are submitted to the Secretary-General of the Consumer Affairs Agency before the product is marketed. However, unlike Foods for Specified Health Uses, the product is not individually pre-approved by the Secretary-General of the Consumer Affairs Agency.
Consumer Affairs Agency in Japan What are "Foods with Function Claims"?
Initiatives in Reliability Assurance
To ensure that our function claims are scientifically valid, in 2014, before the system of "Foods with Function Claims" was launched, we have established the Quality Compliance Section, responsible for validating supporting data and function claims—independently of Group companies and the departments responsible for conducting fundamental research, developing food and beverage products, submitting the Consumer Affairs Agency, and manufacturing and marketing them.
The Quality Compliance Section, while an in-house organization, strictly checks the validity of scientific evidence and labeling from a third-party standpoint throughout the entire process, from material research to product launch and post-launch follow-ups.
Roles Group Companies and Departments Play for Foods with Function Claims
Flow from Fundamental Research for Materials to Post-Launch Follow-Ups
Confirmation of safety and effectiveness
A high level of expertise is required to obtain, investigate, and evaluate scientific evidence regarding the safety and effectiveness of Foods with Function Claims. Therefore, in each process from fundamental research into materials to product development and post-launch follow-ups, each department confirms the safety and effectiveness of the product while demonstrating its expertise.
-
*Press the words Fundamental Research into Materials, Product Development, or Post-Launch Follow-Ups to toggle the display.